Ministry: Surgery prep applicator voluntarily recalled
February 21, 2025
Share this article
Click to view full size
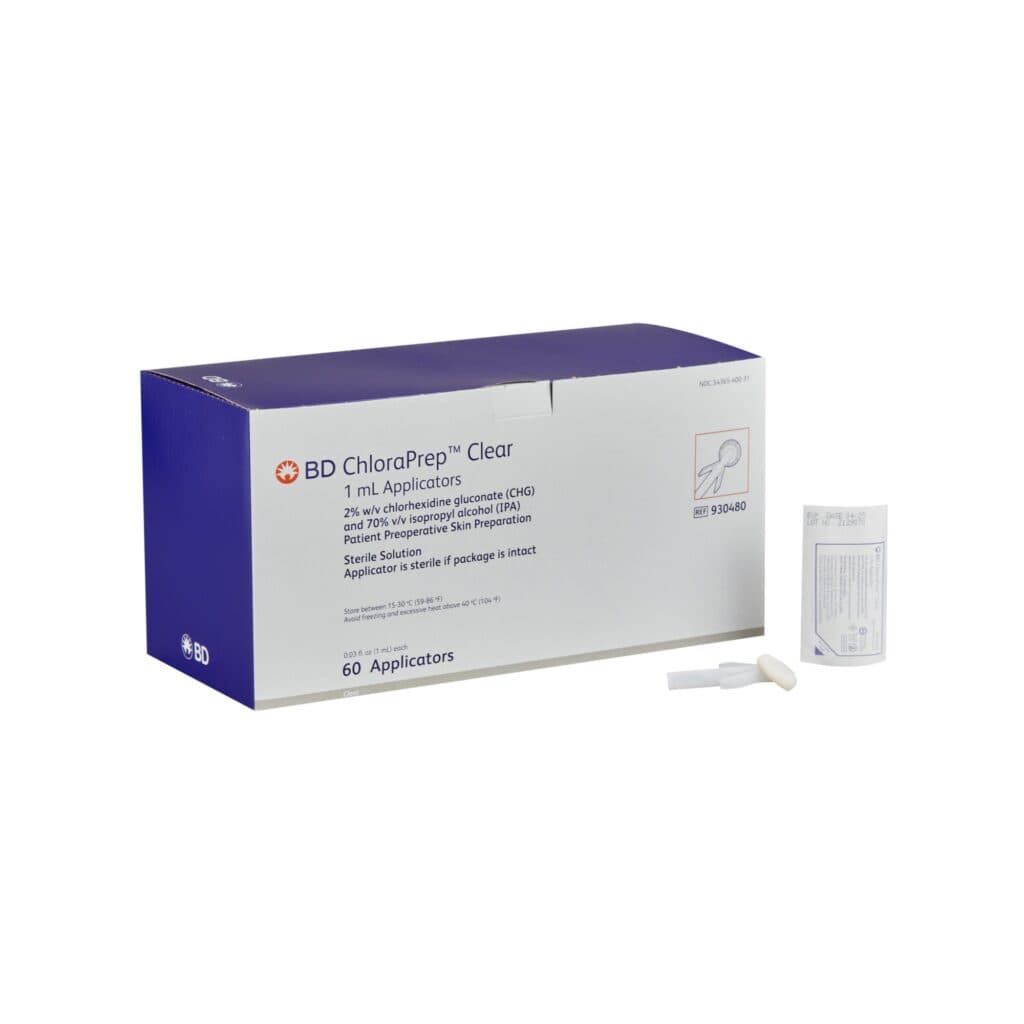
The Ministry of Health says there has been a voluntary recall of one lot of ChloraPrep Clear 1 mL Applicator. These applicators are used for the preparation of a patient’s skin prior to surgery and helps reduce bacteria that can potentially cause skin infections.
In a release, the ministry said the recall, as stated in a release from BD (Becton, Dickinson and Co), is “due to fungal contamination under certain environmental conditions allowing the growth of Aspergillus penicillioides."
The Aspergillus penicillioides within the packaging can contaminate the surface of the applicator and/or gloved hands of the healthcare professional and then consequently the sterile field.
Share this article
Comments (0)
Download our mobile app
The portable companion to gazettE. Get notifications, track read articles, and more. The latest news from Trinidad and Tobago, in one place.





